Blood irradiation indicators
Blood irradiation indicators are used to provide a clear visual check that blood products have passed through an irradiation process. In hospital transfusion services, blood banks, and related clinical environments, they support safer workflow control by helping staff distinguish blood products that have been irradiated from those that have not. Competitor materials consistently position these indicators as process verification tools used in blood safety programmes, particularly where irradiation is part of practice intended to reduce the risk of transfusion-associated graft-versus-host disease.
 Open product: Rad-Sure™ Blood Irradiation Indicators
Open product: Rad-Sure™ Blood Irradiation IndicatorsRad-Sure™ Blood Irradiation Indicators
chemistry: radiochromic film look for the NOT Rad-Sure™ is a blood irradiation indicator that provides… Read more
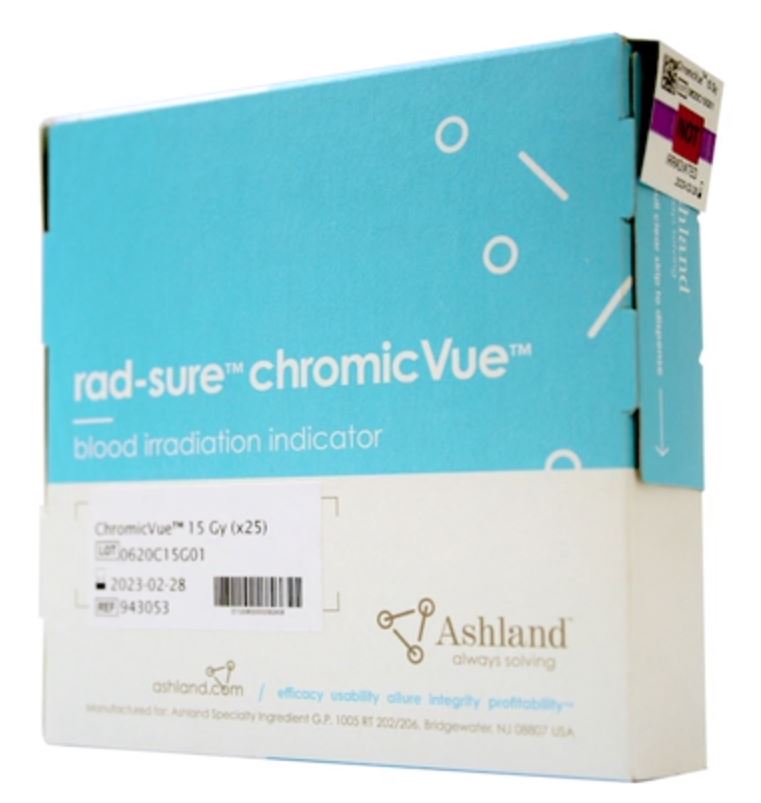
 Open product: Rad-Sure™ ChromicVue™
Open product: Rad-Sure™ ChromicVue™Rad-Sure™ ChromicVue™
Rad-Sure™ ChromicVue™ blood irradiation indicators provide all the dependable features of the traditional Rad-Sure indicators… Read more
For procurement teams and clinical stakeholders, this category matters because the product is simple in form but important in routine documentation, traceability, and day-to-day handling. Buyers need indicators that are easy to read, practical to apply, compatible with the irradiators in use, and suitable for established blood management procedures. PEO Medical supplies blood irradiation indicators from Ashland, supporting healthcare providers and project buyers who need dependable consumables for blood product handling workflows. Ashland states its Rad-Sure indicators are used with gamma and X-ray sources and include features such as room-temperature storage, cGMP alignment and ISBT 128 bar-coded lot numbers.
What is Blood Irradiation Indicators?
Blood irradiation indicators are adhesive process indicators applied to blood products to show, by a visible change, whether the product has been exposed to irradiation. Ashland describes a before-and-after readout in which the indicator initially reads “NOT Irradiated” and, after exposure, the word “NOT” is obscured so the label reads “IRRADIATED”.
These products are not a substitute for a full irradiator quality assurance programme, but they are a practical part of blood safety workflow. Their role is to support visual verification at the unit level, helping staff manage handling, storage, release, and documentation of irradiated blood products within routine practice. RadTag and Ashland both position the category around verification of irradiation for blood products handled by hospitals and blood banks.
How Blood Irradiation Indicators Products Are Used
In practice, a blood irradiation indicator is attached to the blood bag or related product before irradiation. After the irradiation cycle, staff inspect the indicator to confirm that the expected visual change has occurred. This creates a straightforward checkpoint in the workflow and can support segregation of irradiated and non-irradiated units. Ashland specifically notes compatibility with both gamma and X-ray sources, which is relevant for facilities using different types of blood irradiators.
Within healthcare environments, these indicators are most closely associated with transfusion and blood management procedures, but the operational value extends to stock handling, traceability, and internal quality processes. They can help reduce confusion at busy workstations, support documentation practices, and make it easier for teams to confirm processing status during routine handling. RadTag also highlights the importance of verification because operator error, machine error, product placement, and load configuration can affect delivered dose.
Applications of Blood Irradiation Indicators
Hospitals
Hospitals with transfusion services and specialist treatment pathways use blood irradiation indicators to support blood product handling procedures and process visibility.
Radiotherapy
Although not a radiotherapy treatment accessory, this category is relevant to radiotherapy-linked hospital environments where irradiators, oncology pathways, and transfusion support services may sit within the same broader governance framework.
Radiology
Radiology departments are not the primary setting, but procurement teams overseeing multiple radiation-related consumables may review this category alongside other controlled-use products.
Oncology
Oncology services are relevant because irradiated blood products may form part of wider transfusion support pathways for immunocompromised patients. Competitor messaging connects the category to prevention of TA-GvHD through blood irradiation.
Clinics and outpatient centres
Specialist centres with transfusion-linked workflows may require clear, easy-to-manage indicators where blood products are prepared, transferred, or checked.
Diagnostic laboratories
Laboratory and transfusion environments benefit from indicators that fit documented handling procedures and support traceability.
Nuclear Medicine
Nuclear medicine is not the main use case, but hospitals that procure across multiple radiation-related disciplines may include this category within broader controlled consumables sourcing.
Key Features and Capabilities
- Clear visual verification of irradiation status, supporting straightforward interpretation in routine workflows.
- Compatibility with gamma and X-ray irradiation sources, depending on product specification.
- Room-temperature storage for practical stock management.
- Adhesive label format suited to blood bag application.
- Traceability features such as ISBT 128 bar-coded lot numbers on selected products.
- Colour-blind-friendly reading approach on Rad-Sure, reducing ambiguity in interpretation.
- Support for maintenance-efficient workflows because the product is simple to issue, apply, inspect, and document.
- Easy integration into existing blood bank and transfusion service procedures.
Benefits of Using Blood Irradiation Indicators
Using blood irradiation indicators can improve everyday process control in blood handling environments. A clear visual check helps teams confirm product status quickly and supports more efficient movement through irradiation, checking, storage, and release steps. This can be especially helpful in high-throughput settings where consistency matters.
From a procurement perspective, the category offers practical value because it supports standardisation, traceability, and staff usability without adding complex equipment requirements. For clinical teams, it helps maintain clear distinction between processed and unprocessed units. For quality teams, it can contribute to documented workflow control when used as part of broader site procedures. Competitor materials also emphasise the importance of irradiation verification because incorrect delivery conditions can occur in practice.
Choosing the Right Blood Irradiation Indicators
When selecting blood irradiation indicators, buyers should start with intended use and irradiator compatibility. If your facility uses gamma or X-ray blood irradiators, the indicator should be matched to that operating environment and to your internal blood processing protocol. Ashland explicitly states compatibility with gamma and X-ray sources for Rad-Sure.
Other considerations include readability, adhesion, storage conditions, labelling and traceability needs, and alignment with local quality procedures. Procurement teams should also consider whether the product supports the documentation standards used by their blood bank or transfusion service. Where governance requirements are important, buyers may also review manufacturer trust signals such as FDA 510(k), cGMP references, ISBT 128 labelling features, or ISO 9001 quality management claims, depending on the supplier and market.
Our Blood Irradiation Indicators Solutions
PEO Medical offers blood irradiation indicators from Ashland, a recognised manufacturer in blood safety consumables. Ashland states that Rad-Sure blood irradiation indicators provide positive visual verification of irradiation at the minimum specified dose and have been used as a standard product in this category for more than 25 years. The range is manufactured using Gafchromic film and is positioned for use with both gamma and X-ray blood irradiation sources.
For buyers, this makes the category suitable for hospitals, blood banks, and transfusion services seeking a practical, easy-to-read verification label from an established supplier. PEO Medical can help customers align product choice with site workflows, purchasing needs, and wider medical equipment procurement plans.
Why Choose PEO Medical
PEO Medical supports hospitals, distributors, ministries of health, NGOs, and healthcare project buyers with specialist product sourcing across radiotherapy, imaging, radiology, nuclear medicine, laboratory, dental, and veterinary sectors. Within that broader supply capability, blood irradiation indicators fit naturally into controlled clinical consumables procurement.
Choosing PEO Medical means working with a supplier that understands healthcare purchasing requirements, international delivery coordination, and the need for clear product selection support. We help buyers identify suitable solutions, compare product fit against operational requirements, and streamline procurement for both public and private healthcare settings.
Conclusion
Blood irradiation indicators are a focused but important product category for transfusion services and blood handling environments. They help teams verify irradiation status visually, support traceability, and contribute to safer, clearer workflow control when blood products are processed for clinical use. Competitor pages make clear that the category is closely linked to blood safety, irradiation verification, and compatibility with established irradiator platforms.
PEO Medical supplies Ashland blood irradiation indicators for healthcare organisations that need dependable consumables for blood management procedures. Explore the products in this category or contact PEO Medical for support in selecting the right option for your facility.